
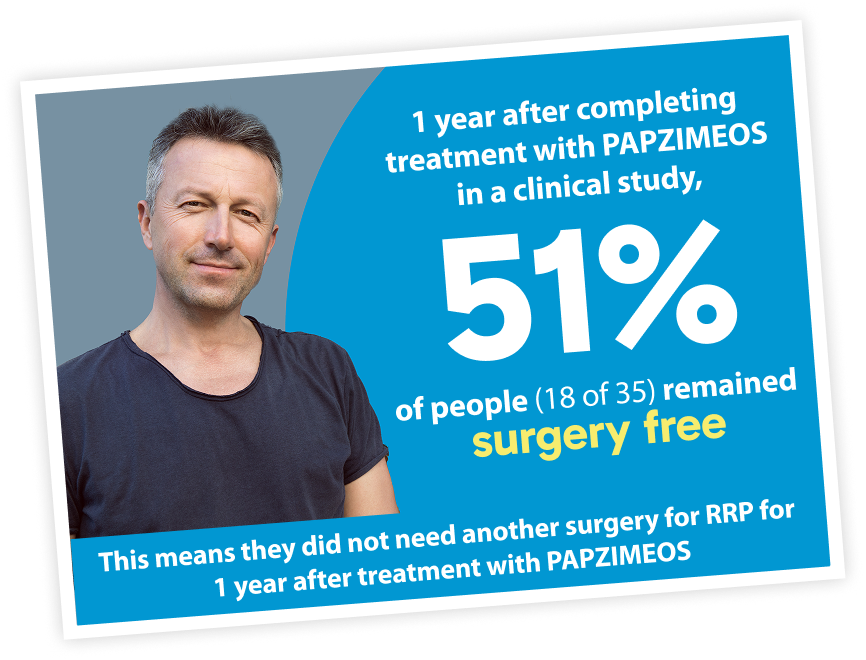
PAPZIMEOS may give patients the chance to have more surgery-free time
PAPZIMEOS is recommended as a first-line treatment for newly diagnosed adult patients with recurrent respiratory papillomatosis (RRP).
Not an actual patient.

Not an actual patient.
OVER HALF HAD
0 surgeries
People with RRP went from having at least 3 surgeries per year before taking PAPZIMEOS to an average of 0 surgeries in the year after finishing treatment
MOST PATIENTS THAT HAD
0 SURGERIES AT YEAR 1 WERE
STILL
SURGERY
FREE
AFTER 2 YEARS
MOST patients that were surgery free after 2 years were
STILL
SURGERY
FREE
AFTER 3 YEARS
14 of 18 patients remained surgery free after 3 years of follow-up. One patient has not yet reached 3 years of follow-up
In the safety evaluation of 38 patients, side effects included pain, redness, or swelling where the shot was given, feeling tired, chills, fever, muscle aches, feeling sick (nausea), headache, increased heart rate, diarrhea, vomiting, and sweating a lot.
THERE WERE 70% FEWER SURGERIES OVERALL 1 YEAR AFTER TREATMENT WITH PAPZIMEOS.
In the year before treatment with PAPZIMEOS, there were a total 151 surgeries for all 35 patients in the trial. In the year after completing treatment with PAPZIMEOS, there were a total of 45 surgeries in some of the patients.
The most common side effects for PAPZIMEOS included*:
- Pain, redness, or swelling where the shot was given
- Feeling tired
- Chills
- Fever
- Muscle aches
- Nausea (feeling sick)
- Headache
- Increased heart rate
- Diarrhea
- Vomiting
- Sweating a lot
*Safety evaluation included all 38 people with RRP studied who received PAPZIMEOS. 35 people in the clinical study received the recommended dose of PAPZIMEOS.
No patients in the clinical trial had to stop taking PAPZIMEOS early due to side effects
Most adverse reactions happened after the first dose of PAPZIMEOS and usually lasted 1-3 days.
In people who took PAPZIMEOS, the most common adverse reactions were mild to moderate and mostly resolved on their own.
These are not all of the possible side effects of PAPZIMEOS. Call your healthcare provider for medical advice about side effects. You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088. You may also report side effects to Precigen, Inc. at 1-855-PGE-NRRP (1-855-743-6777).
Experts supported by the RRP Foundation
recommend PAPZIMEOS for adults with RRP who want to break the cycle and reduce their number of surgeries.
The goal of RRP treatment has evolved its focus from managing symptoms to helping patients have fewer surgeries, which may help patients avoid potential complications due to additional surgeries.
Not an actual patient.

